Printable Electronics Reference Devices and Materials
Release status: Experimental
| Description | Printable Electronics Reference Devices and Materials
|
| License | |
| Author | |
| Contributors | |
| Based-on | |
| Categories | |
| CAD Models | |
| External Link |
Contents
- 1 Description:
- 2 The Teflon Wick design for the Micron pen:
- 3 Field Effect Transistor Patterns:
- 4 Bistable Inverter:
- 5 Materials:
- 6 Silver Ink:
- 7 Commercial Organic Semi-Conductors:
- 8 Zero-Cost Organic Semiconductors:
- 9 Dielectrics:
- 10 Substrates:
- 11 Resistor:
- 12 Glow in the dark:
- 13 Capacitor:
- 14 Surface Mount Motherboard Design for discrete components:
Description:
This is a home for a set of reference devices and materials for printable electronics.
The Teflon Wick design for the Micron pen:
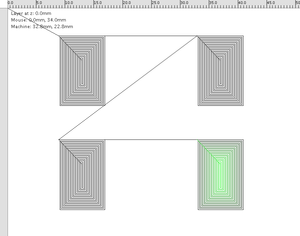
Field Effect Transistor Patterns:
Gate layer:
Dielectric layer:
Semi-conductor layer:
Source-Drain layer:
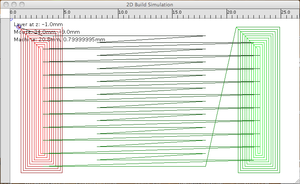
Fet CLUE session 12-09-10:
The gcode is machine specific:
File:Fet10bistableInverterGate.gcode.txt
File:Fet10bistableInverterInsulator.gcode.txt
File:Fet10bistableInverterSemiconductor.gcode.txt
File:Fet10bistableInverterSourceDrain.gcode.txt
File:Fet10bistableInverterRoute.gcode.txt
File:Fet10bistableInverterTextCH.gcode.txt
The Common-Lisp script that generates the gcode:
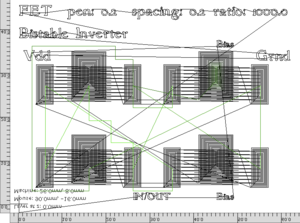
Bistable Inverter:
Materials:
There is a direct relation between non-toxicity and ease of printing.
- Less toxic, less cost.
- With the same material different solvents will have unique morphologies.
Silver Ink:
Features:
- Water based
- While printing it is pretty close to non-toxic. If the tool head touches you while printing you probably won't die.
- cold valveless tool head
- With the right particle size cure temperatures can be below plastic extrusion temps.
Design:
Previously, we demonstrated that sub-micron silver ink could be printed into circuits and active devices with a modified Micron pen.
After printing, the ink particles were sintered on a hot plate to increase the conductivity.
The process worked well, but the ink was not readily available. Here is a possible method to fabricate the metallic ink. This would allow a cold process metal printing of circuits, devices and 3D structures.
Ionic Salt Crystals:
A metal and an acid can form an ionic salt that is water soluble.
The metal can be arranged in solution.
Then the salt can be reduced back into the metal with a reducing agent.
This might be a general way to deliver a range of metals via a water solution.
Or the particles can be reduced in the solution to yield a bunch of small particles. The colloid of particles would then be put in the pen.
Silver + Nitric Acid => Silver Nitrate(stains skin) + water + Nitrogen Oxide(a toxic gas)
3 Ag + 4 HNO3 → 3 AgNO3 + 2 H2O + NO
A solution of small silver particles can then be made by mixing the Silver Nitrate with a reducer such as Ascorbic Acid or Sodium Citrate. This transfers an electron back to the silver ion.
Silver Nitrate + Ascorbic Acid = Silver + DHA + Nitric Acid
2AgNO3 + C6H8O6 = 2Ag + C6H6O6 + 2HNO3.
To prevent the small particles from clumping or redisolving, a stabilizer or “capping agent” is added such as Polyacrylic Acid (PAA).
PAA + Diethanolamine(DEA) or polyvinylpyrrolidinone (PVP) or Sometimes a dispersing surfactant is used instead.
PAA is the absorbent powder in diapers and is listed as an irritant.
This solution is then heated mildly on a hot plate from 1hr to 24hrs.
I guess this is mainly for brownian motion.
Next, Ethanol(grain alcohol) which is less polar than water can be added to the water solution which will allow the particles to attract each other and precipitate out.
The particles can be further concentrated by spinning them.
Viscosity:
Viscosity increases with the concentration of particles.
Solutions of 50% silver particles with a small amount of a viscosity additive were reported to have a viscosity of 0.1-1.0cP.
Previously successful inkjet inks had a listed viscosity of 12 cP.
Normal drawing inks are near the viscosity of water 0.894 cP.
So target concentrations of 50% silver particles by weight should work.
Notables:
Printed silver process by Thomas Edison.
(A)Silver nitrate (metal salt)
(B)reduced with printed vitamin C
Edison process was redone 2007 with ink-jets by Seyed Bidoki and Matthew Clark at University of Leeds.
Ink-jet fabrication of electronic components
S M Bidoki, D M Lewis, M Clark, A Vakorov, P A Millner and D McGorman
NerdRage: Making Silver Nitrate
Pen-on-Paper Flexible Electronics
Analisa Russo, Bok Yeop Ahn, Jacob J. Adams, Eric B. Duoss, Jennifer T. Bernhard, and Jennifer A. Lewis
Wikipedia Ethanol precipitation
Preparation and the mechanisms of formation of silver particles of different morphologies in homogeneous solutions, Lorenza Suber, Ivan Sondi, Egon Matijevic, Dan V. Goia
3D printed silver-ink devices were printed by ink-jet in 2002:
Fuller, S. B., E. J. Wilhelm, et al. "Ink-jet printed nanoparticle microelectromechanical systems." (2002) Journal of Microelectromechanical Systems 11(1): 54-60.
Commercial Organic Semi-Conductors:
TIPS-Pentacene
P3HT
Polypyrrole (PPy)
Electronic Conduction in Polymers: The Chemical Structure of Polypyrrole, by R. McNeil (1963) (suggested by Arturo and others)
Vat Yellow 1
http://en.wikipedia.org/wiki/Vat_dye
http://en.wikipedia.org/wiki/Vat_Yellow_4
Perylene-diimide
Zero-Cost Organic Semiconductors:
Nature is full of amazing electrical phenomena!
The Class of Natural Pigments:
They are selected by nature to absorb light.
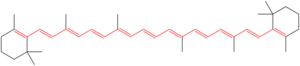
Beta-carotene:
Mobility 4×10^(−4) cm^2/V/s
Number Check: 7 orders of magnitude lower than the electron mobility in silicon
Notables:
Lycopene has a striking resemblance to Polyacetylene and Beta-carotene.
http://www.nobelprize.org/nobel_prizes/chemistry/laureates/2000/public.html
Biocompatible-ingestible electronic circuits:
Biocompatible and Biodegradable Materials for Organic Field-Effect Transistors, by Mihai Irimia-Vladu
Exotic materials for bio-organic electronics, by Mihai Irimia-Vladu (recommended by Arturo)
Chlorophyll:
Dielectrics:
PVP
PMMA
The CP-1 test suggests art-store grade dielectrics are probably not good enough.
mrkim: The successful use of PVP and PMMA makes me doubt that result.
Glucose
Substrates:
Glass microscope slides
Resistor:
Graphite Pencil
Carbon particle "Wire Glue" ink.
Glow in the dark:
zinc sulfide
Capacitor:
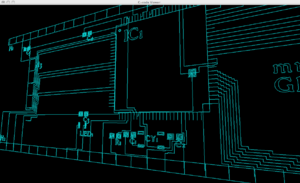
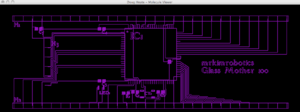
Surface Mount Motherboard Design for discrete components:
Components:
Atmega1280